Here’s Where To Buy At-Home COVID-19 Tests
The FDA issued emergency use authorization to a prescription-based at-home COVID-19 test that provides results in less than 30 minutes.

As we navigate another year of the COVID-19 pandemic, surges and variants have become an uncomfortable part of everyday life. The Omicron variant, or B.1.1.529, is currently the dominant strain in the United States, according to the Centers for Disease Control and Prevention (CDC). In early December, the Delta variant was responsible for most U.S. COVID-19 cases.
Experts in This Article
professor of medicine at Harvard Medical School
infectious disease epidemiologist and public health professor at the University of Nevada, Las Vegas
Each variant brings confusion and adjustments—the Omicron variant is two to three times more contagious than Delta, which means gathering unmasked and indoors is more precarious than ever. It’s also worth noting that viral mutations are natural: Viruses, including SARS-CoV-2, continue to survive because they mutate, Karen Edwards, PhD, a professor and chair of the department of epidemiology at the University of California, previously told Well+Good.
“When a virus like [SARS-CoV-2] is circulating in a population—the more times it basically replicates itself—the more opportunities there are for mistakes,” Dr. Edwards said. “Those mistakes are what we call ‘mutations.” Vaccinations and booster shots remain the most effective public health measurements. They protect the population from severe illness and reduce opportunities for mutation, but COVID-19 testing is an instrumental tool for slowing community spread.
You’re likely familiar with two types of COVID-19 tests: “The PCR test is looking for the genetic material,” Brian Labus, PhD, MPH, an infectious disease epidemiologist and public health professor at the University of Nevada, Las Vegas, previously told Well+Good. “The antigen test is looking for the proteins, basically the structure of that virus.”
PCR tests are the most accurate form of COVID-19 screening available to the public, but they are costly and typically take a few days to yield results. At-home rapid antigen tests aren’t as sensitive as PCR tests, but they are very effective at screening for contagious COVID-19 cases. A 2021 pre-print study looked at 250 adults with suspected COVID-19 and found that rapid antigen tests were 97 percent likely to prove someone is both positive for COVID-19 and infectious. This data suggests that screening people with antigen tests is a good approach to prevent COVID-19 outbreaks because the results are, well, rapid. “The advantage of at-home tests is that they are fast,” Dr. Edwards explained. “You get the results immediately, in 10 to 15 minutes, for example.”
This information isn’t exactly groundbreaking—most people know how helpful regular testing can be. However, finding at-home test kits is a bit more challenging because they are frequently sold out, but we’ve compiled a list of 11 kits approved by the U.S. Food and Drug Administration–and stores that might have them in stock.

BinaxNOW COVID-19 Antigen Self Test — $14.00

CareStart Covid-19 Antigen Home Test Kit — $30.00

InteliSwab COVID-19 Rapid Antigen Home Test Kit — $14.00

ellume COVID-19 Rapid Antigen Home Test — $26.00
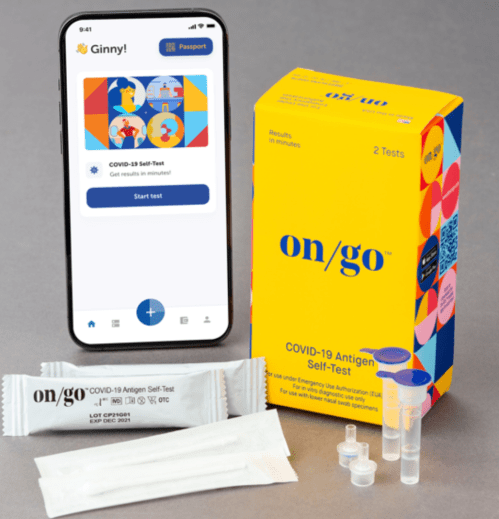
On/Go at-Home COVID-19 Rapid Antigen Self-Test — $30.00
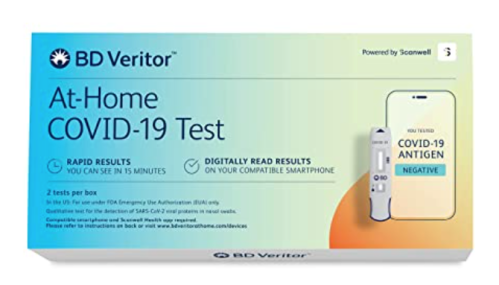
BD Veritor at-Home COVID-19 Digital Test Kit — $34.00
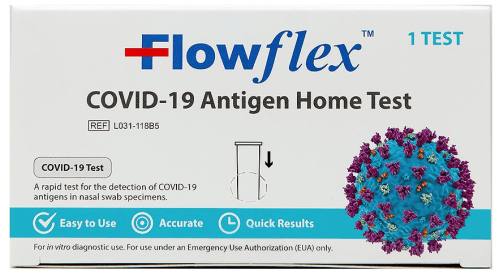
FlowFlex COVID-19 Antigen Rapid Home Test Kit — $10.00

QuickVue Rapid At-Home COVID-19 Antigen Test Kit — $24.00

Lucira Check It COVID-19 Test Kit — $89.00

Cue Health — $225.00
Sign up for the Well+Good SHOP Newsletter
Get exclusive deals on wellness, beauty, fitness, and food products that have been hand-picked by our editors.
Got it, you've been added to our email list.







